Stray-CATS
Stroke treatment goes personalized: Gaining added diagnostic yield by computer-assisted treatment selection (STRAY-CATS)
There is now class I evidence that mechanical thrombectomy is a safe and effective therapy within an estimated time frame of 6 h after stroke onset. Seven prospective studies have recently evidenced the superiority in proximal vessel occlusions (MR-CLEAN, ESCAPE, EXTEND-IA, SWIFT-PRIME, REVASCAT, THRACE, and THERAPY). Mechanical thrombectomy has thus become the treatment option of choice to achieve an early and sustained revascularization of proximally occluded vessels in tertiary care stroke centers. These studies have illustrated that mechanical thrombectomy using stent retrievers is a highly effective treatment with low complication rates. However, due to the narrow inclusion criteria and limed time window for treatment, the studies do not provide a guide for the majority of clinical patients who do not match inclusion criteria such as for example presentation beyond the time window of 6 hours of or with large infarct core at initial imaging. The aim of decision making for endovascular stroke treatment is to maximized the number of patients who will benefit from recanalization and minimized the patients will futile recanalization after endovascular stroke treatment. To facilitate this, an individualized prediction model of the neurological outcome in case of recanalization compared to the natural history is necessary.These studies have highlighted the potential to select patients based on the size of the penumbra and ischemic core, but have raised new questions to be solved: “can we predict a benefit in tissue survival if mechanical thrombectomy is successfully applied compared to the natural course of disease”. This question is strongly dependent on the presence of sufficient vs. insufficient collaterals.” The availability of a fast, safe, reproducible and reliable information about the expected tissue salvage would allow us not only to select patients that would benefit from mechanical thrombectomy, it would also permit selection of patients for revascularization in a more flexible time window that might exceed 6 h if sufficient collateral flow enables sustained tissue survival.
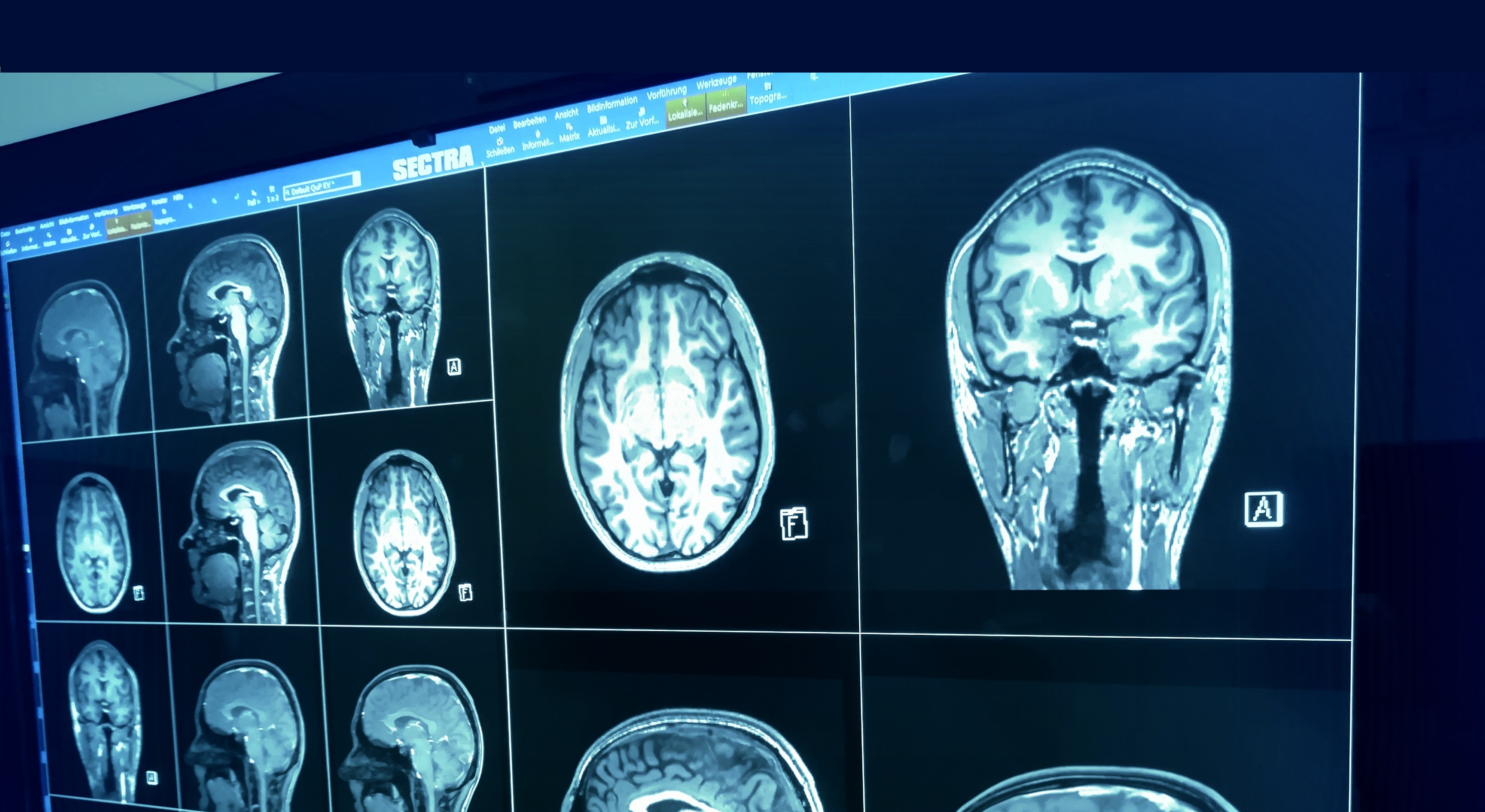
It is essential that indicators for further success of endovascular therapy must be calculated as soon after admission as possible, in order to save as much of the brain tissue as possible: “time is brain”. In this regard, computer-assisted and automated tissue segmentation of the infarct core and salvageable penumbra using compound information from multimodal MRI offers a novel and robust standardized solution to this problem. We have recently devised an algorithm for “Fully Automated Stroke Tissue Estimation using Random Forest Classifiers” (FASTER), that enables nonlinear estimation models to determine voxels as either at risk for infarction or being potentially salvageable based on a compound feature extraction from multimodal MRI data. FASTER provides, given an individual patient case with open outcome, a comparison between outcome predictions in case of a successful vs. an unsuccessful reperfusion. FASTER gives a more precise delineation of tissue-at-risk than that given by an expert rater using linear thresholded maps, and operates within a timeframe of 6-10 minutes after calculation of the perfusion maps. Within a retrospective data analysis that included overall 100 patients, lesion volume predicted by FASTER in the case of a poor response to therapy was significantly correlated with the final lesion volume in test cases having TICI score 1-2a.
Within the STRAY-CATS project, we aim to transform the FASTER algorithm into a clinical applicable tool for computer-assisted treatment selection. We thus aim to extend the nonlinear estimation model by i) linking individual prediction maps to lesion topography, in order to correlate individual lesion load in eloquent brain areas with subsequent outcome, and ii) integrating quantification of penumbral collateral flow to better identify the potentially recoverable population.
The ultimate goal of the project is to provide a system capable of assessing patient risk in acute stroke, based on imaging features and quantification of collateral flow derived from pre-interventional imaging. This information will benefit stroke patients, by providing neuroradiologists and stroke neurologists with a fast, accurate, repeatable interpretation of multiple disparate information sources. Evidence from our system will enable better treatment selection by refining the estimation of irreversibly damaged vs. potentially salvageable penumbral tissue and its respective lesion load on eloquent areas of the brain. An accurate computer-assisted treatment selection module offers the potential to select patients with stable collaterals and preserved penumbral from even beyond the currently recommended window of 6 hours after stroke onset.
Such a tool require to be robust to differences in vendor, field strength and sequence parameters. To this end, we will build on our existing predictive models of stroke outcome, extending the training data for the prediction by using pooled datasets, incorporating imaging data from patients that underwent intra-arterial thrombolysis or thrombectomy within an established transatlantic framework between the stroke centers of the Inselspital, University of Bern and the Westwood Clinics, University of California, Los Angeles and their associates. For the purposes of testing our methodology, we will utilize data from the STAR trial and the upcoming SWIFT direct trial: these datasets will provide a broad basis for the performance of computer-assisted treatment selection in a variety of settings. We will pay particular attention to cases beyond the established 6 hour time window, performing a subgroup analysis on these cases to establish that automated tissue analysis can be used for treatment selection in the most difficult cases.
We will make use of an existing cooperation with medical imaging developers (OLEA Medical, La Ciotat France) to accelerate the technology transfer into a clinical workflow by encapsulating the developments into a clinical software module.
Responsible persons:
Jan Gralla